The rCBF(Alpert) model is used for calculating parametric perfusion maps from dynamic H215O brain PET images. A 1-tissue compartment model is applied
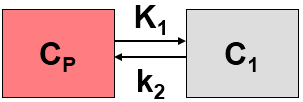
with operational equation
![]()
where K1 represents perfusion F, and k2 equals perfusion divided by the particion coefficient of water p. Alpert et al. [1] introduce weighting functions w1(t) and w2(t) which are multiplied with both sides of the equation above. Resolving the equation multiplied by w1(t) yields
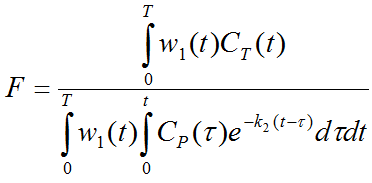
Dividing both multiplied equations results in an expression which only contains k2.
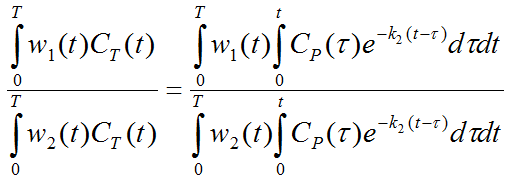
Based on the latter two equations the following methodology for calculating perfusion was developed [1]:
1.The weighting functions are defined as w1(t)=1 and w2(t)=t.
2.Since the input curve CP(t) and the physiologic range of k2 are known (the particion coefficient is more or less constant at a value of about 0.9), the right side of the third equation can be evaluated and tabulated for a pre-defined k2-range. This table was called the R-Table and contains the evaluated ratios ri for all k2i.
3.For a tissue curve CT(t) to analyze, the left side of the third equation is evaluated, resulting a value r. It is then looked up in the R-Table which value of k2 results in the ri value closest to r.
4.Using the obtained k2, the second equation can be evaluated, resulting in the perfusion value F.
Acquisition and Data Requirements
Image Data |
A dynamic PET data set representing the measurements of brain activity after the injection of a H215O bolus. |
Blood Data |
Blood activity sampled at a peripheral artery from the time of injection until about 30 seconds post acquisition. The sampling past the end of the PET acquisition is required because of the delayed arrival of blood in an external radioactivity counter. |
TAC for Blood Preprocessing |
A tissue TAC is required to fit a compartment model during blood preprocessing. Candidate TACs would be the imported system count rate, or the average activity calculated in a brain VOI as shown in the example below. |
Blood Preprocessing
The instantaneous blood activity during the acquisition is needed as the input curve of the perfusion model. However, because blood activity is monitored with an external device, the measured activity is distorted relative to the activity arriving in the brain by two effects: a relative time delay, and a broadening of the activity shape (bolus dispersion). For accurate rCBF measurements the delay and dispersion of the blood measurements must therefore be corrected for.
The blood preprocessing step of the rCBF (Alpert) model applies the methodology developed by Meyer et al. [2] for the delay and dispersion correction. It fits a 1-tissue compartment model including a delay and a dispersion parameter to a tissue time-activity curve provided. The blood curve is then explicitly shifted by the found delay, and deconvolved with the exponential dispersion.
A typical definition provided with the example H215O data set is shown below.
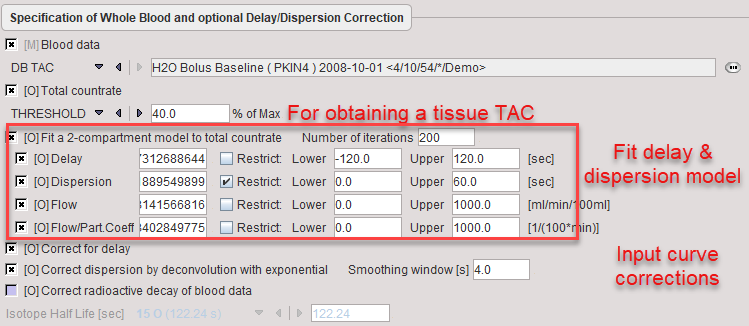
The measured blood activity is read from the file H2O Bolus Baseline in the database. The brain TAC which is used for fitting the 1-tissue compartment model is derived from a 40% THRESHOLD. Alternatively, a user-defined VOI could be used.
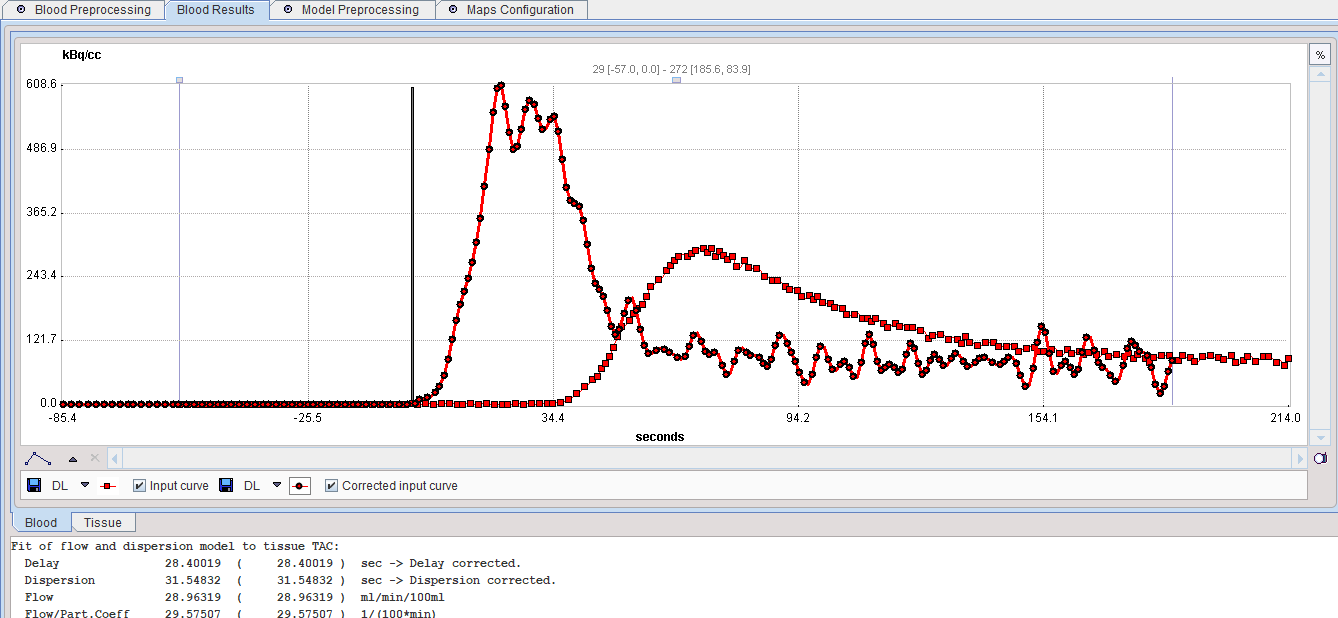
After the blood preprocessing has been performed, the result is shown in the Result panel. The parameters resulting from the fit are shown in the Info area. There are two sub-panels, one showing the corrected blood and the other showing the fit to the TAC.
The Blood curve area shows the original blood measurements and the input curve after correction for the Delay (28.4 sec) and Dispersion (31.5 sec). The Corrected blood data is used for the further calculations.
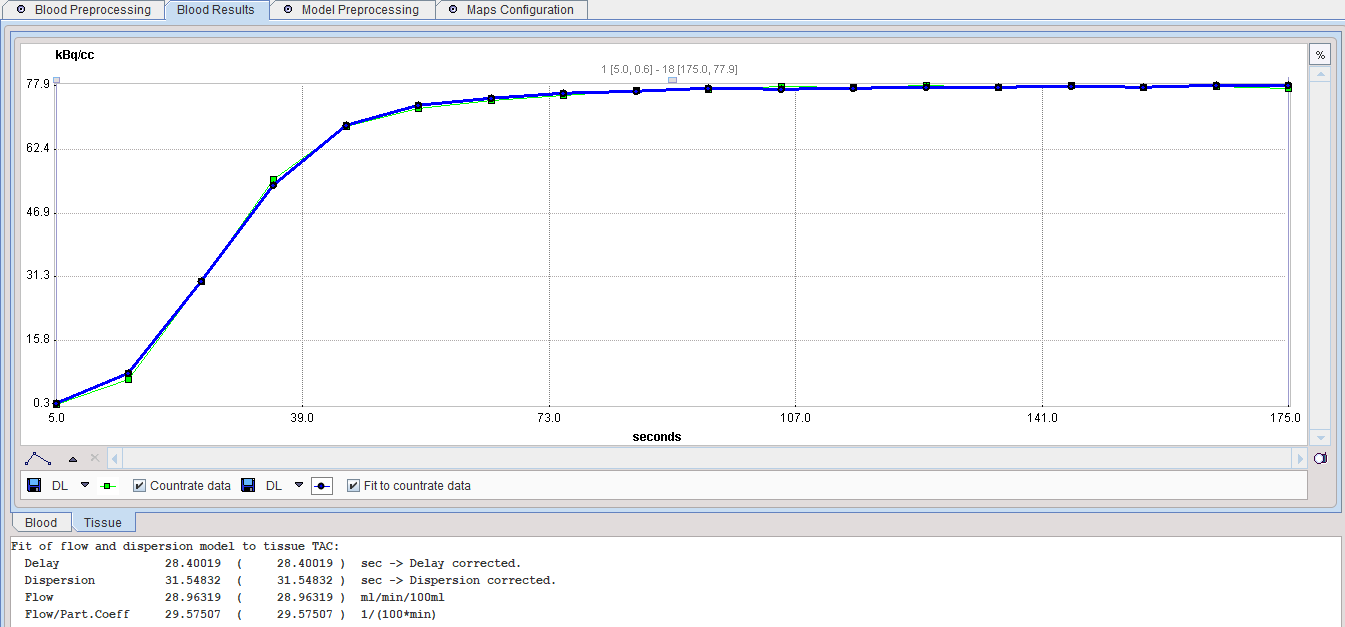
The TAC panel should also be inspected to verify that the fit was successful.
Some published results regarding the dispersion parameter:
•Net internal dispersion due to the longer distance left ventricle-radialis than left ventricle-brain: 4sec [8], 4-6sec [5].
•External dispersion in the sampling tubes: 0.5sec (10mm catheter, 0.5mm inner diameter, no three-way taps, 10ml/min) [5]; 10-12sec, 5-6sec, 1-2sec for 5,10, 20 ml/min (40mm catheter, 1.0mm inner diameter, three-way taps) [8].
Model Preprocessing
Model preprocessing for Alpert's time-weighted integral method consists of calculating look-up tables of the model response within a range of f/p values (f = Flow, p = partition coefficient; f/p = k2 of the 1-tissue compartment model). The f/p range and the number of tabulated values must be specified in the preprocessing dialog as illustrated below. Normally there is no need for changing any of the input parameters.
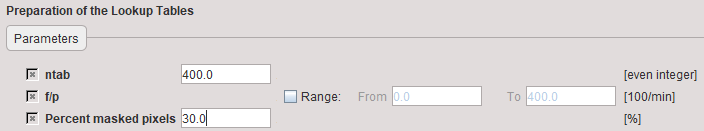
ntab |
Number of pre-calculated values in the look-up table. Should be an even number. |
f /p |
Flow/partition coefficient ("k" in Alpert's paper). The Lower and Upper bound should be specified reasonably, otherwise there will not be a meaningful result. This range is used for setting up Alpert's "r-table". |
Percent masked pixels |
Exclude the specified percentage of pixels based on histogram analysis of integrated signal energy. Not applied in the presence of a defined mask. |
Model Configuration
The main result of the processing is the rCBF value f. Therefore it should be enabled in the Model dialog. f/p and p can also be mapped if needed.
![]()
f |
Regional brain perfusion in [ml/min/100ml]. It is calculated for each pixel by a table look-up procedure (Alpert's eq. 5). Reasonable values are about 30 (white matter) and 60 (gray matter) ml/min/100ml [11]. |
f/p |
The f/p value found in the table look-up procedure and used to calculate f. |
p |
Partition coefficient or distribution volume of water in brain. It is calculated by dividing the two previously calculated parameters. Reasonable values are around 0.9. |
Reference
1.Alpert NM, Eriksson L, Chang JY, Bergstrom M, Litton JE, Correia JA, Bohm C, Ackerman RH, Taveras JM: Strategy for the measurement of regional cerebral blood flow using short-lived tracers and emission tomography. J Cereb Blood Flow Metab 1984, 4(1):28-34. DOI
2.Meyer E: Simultaneous correction for tracer arrival delay and dispersion in CBF measurements by the H215O autoradiographic method and dynamic PET. J Nucl Med 1989, 30(6):1069-1078.